Sign Up Now!
OrganoidXplore™: The first large panel based Organoid screening service
OrganoidXplore™
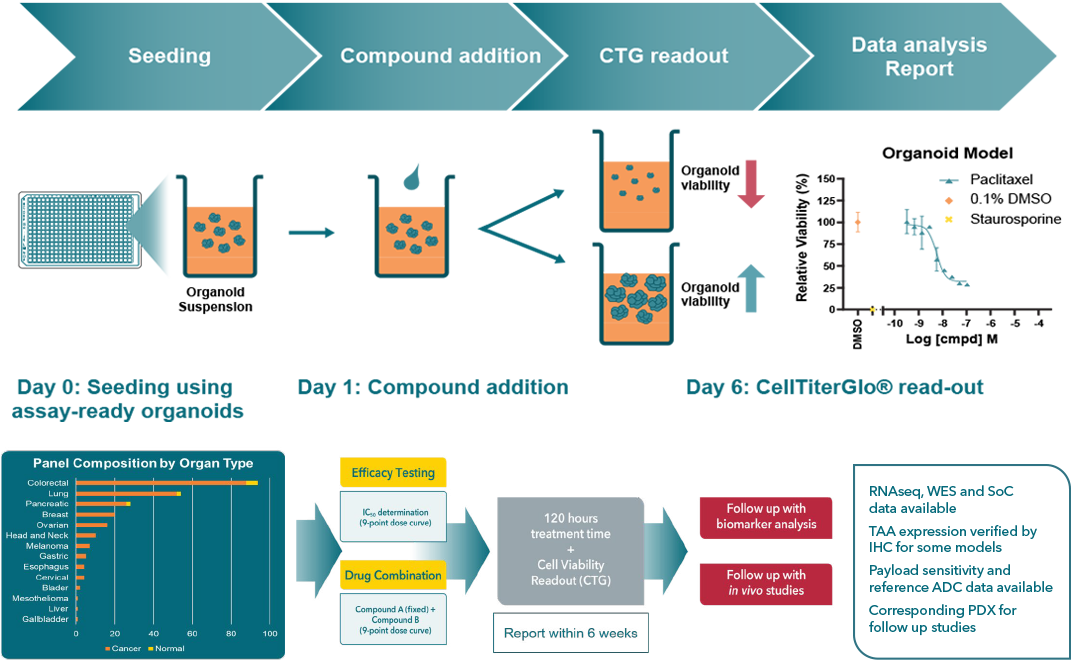
247 fully characterized organoid models, results in 4-6 weeks. Benefit from a streamlined 5-day incubation time, facilitating timely results and accelerating your research timelines. Our automated workflow ensures accuracy and reproducibility across runs with state-of-the-art liquid handlers, delivering consistent Z’-factor >0.2.
Limited-Time Promotional Offer
Exclusive Pricing Benefits:
- 30% discount per model, per compound
- 50% cost share opportunity on Standard of Care compounds
- Rolling submissions accepted
Key Dates:
- Registration Deadline: June 5, 2026
- Screen Execution: June 15, 2026
- Results Delivery: 6 weeks post-execution
Available in Netherlands or China facilities
Secure Your Spot Now
Don't miss this limited-time opportunity. Registration closes June 5th, 2026.
Why Choose OrganoidXplore™?
| Key Area | Noteworthy Benefits |
|---|---|
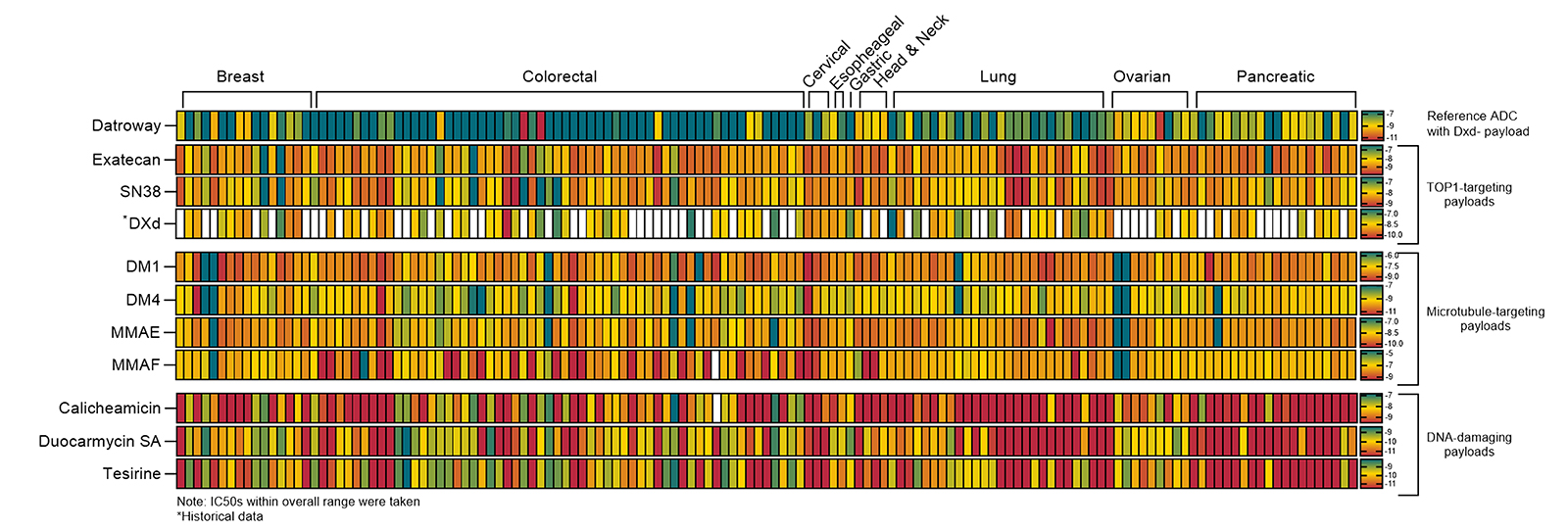
| Unmatched Scale & Diversity | Access 247 well-characterized PDO and PDXO models spanning 14 cancer types with diverse genetic backgrounds including KRAS, BRAF, EGFR, BRCA1/2, and other clinically relevant mutations. |
| Multiple Panel Options | Choose from Large Panel Screen (247 models), KRAS Panel, Lung Panel, CRC Panel, or Flexible Panel (20+ custom model selection). All panels eligible for 30% promotional discount. |
| 14 Cancer Type Models | Explore and test efficacy across colorectal, lung, pancreatic, breast, ovarian, gastric, head and neck, cervical, esophagus, bladder, gallbladder, liver, melanoma, and mesothelioma. |
| Rich Molecular Profiling | Every model includes RNAseq, WES data, and TAA expression profiling with 60+ validated IHC assays—essential for ADC development and patient stratification. |
| Comprehensive Analysis | Investigate off-target effects with matched normal/disease pairs, enabling thorough comparative safety analysis. |
| Seamless In Vivo Transition | Access corresponding PDX models from our extensive biobank to validate organoid findings and advance lead candidates with confidence. Proven high correlation between PDXO and PDX response. |
| Clinical Relevance | Organoid predictions correlate with patient clinical response: 84-100% sensitivity, 69-100% specificity across multiple cancer types in published studies. |
In-Depth Customer Report
Receive a comprehensive report featuring:
- 9-point dose-response curves or 9x1 matrix treatment for every model tested
- IC50 and AUC values with confidence intervals
- Brightfield imaging at day 0 and day 5 for quality
- Study design and methodology details
- Control performance metrics (Z-factor, CV%)
- Raw data files for independent analysis
- Model characterization data (mutations, expression profiles)
- Comparison to SOC agents (when applicable)
Flexible Add-On Services to Maximize Your Investment
Biomarker Identification & Patient Stratification: Integrate differential expression analysis between responders and non-responders. Leverage our RNAseq and WES data to identify predictive biomarkers and stratify patient populations for clinical development.
Mechanism of Action Studies: Opt for high-content imaging to visualize ADC penetration, measure bystander killing effects, and assess antibody binding, internalization, and co-localization—critical for ADC optimization.
Drug Combination Studies: Explore enhanced therapeutic potential through combination studies, opening new avenues for synergistic treatment strategies.
Matched In Vivo Validation: Seamlessly transition to efficacy studies with corresponding PDX models from our biobank. Validate organoid predictions with PK/PD profiling and in vivo efficacy to support clinical translation.
ADC Stability Analysis: Assess stability in plasma/blood via LC-MS and ELISA. Monitor DAR with LC-HRMS over time and evaluate payload release under physiological conditions.
Proven Success Across Therapeutic Modalities
Targeted Therapies: Validated with KRAS G12C inhibitors (Sotorasib), RAS-ON inhibitors (Daraxonrasib), and molecular glues (CC-885)
ADC Development: Comprehensive characterization with TOP1-targeting ADCs (Datopotamab deruxtecan, Trastuzumab deruxtecan) and 10+ common payloads
Chemotherapies: Biomarker discovery and patient stratification across diverse genetic backgrounds