Humanized PBMC Mouse Models
Evaluate your novel immuno-oncology agents in our platform of transient human immunity
Looking For a Simplified Approach to Humanization?
Humanized PBMC mouse models offer a unique opportunity to study immunotherapeutics within a human tumor microenvironment, while providing a simple alternative to the full stem cell reconstitution approach.
The platform is created by mixing human peripheral blood mononucleated cells (PBMC) with Cell Line Derived Xenograft models. The PBMC injection route can be subcutaneous (where the PBMC are admixed with tumor cells) or intravenously or intraperitoneally, followed by tumor cell inoculation.
Our humanized models have been fully validated to show that the partial reconstitution of human hemopoiesis occurs, and in specific models the significant replenishment of the human T cell population coincides with the appearance of immunotherapeutic efficacy.
Multiple Model Types Already Available for Efficacy Testing...Can't See What You Need? Just Ask
We already provide a wide range of validated models, which can be expanded as required to fit our clients needs. Our current platform encompasses models of:
- Breast cancer (BT-474)
- Colorectal cancer (COLO 205, HCT 116, SW480)
- Lung cancer (HCC827)
- Lymphoma (KARPAS-299, Jeko-1)
- Melanoma (A375)
The selection of new models for development is made easy through our curated online cell line and Cell Line Derived Xenograft database , and our powerful new search engine (which searches models from all our Oncology databases)
Our Cell Line Derived Xenograft models are fully characterized and collated with their genomic annotation, allowing rapid selection of models based on genetic characteristics such as overexpressing antigens of interest.
One Platform, Many Uses
The humanized PBMC platform is available for a variety of immuno-oncology applications, and our experience includes:
- CD19, HER2, EGFR, and EpCAM BiTE®-like antibodies
- Evaluation of immune checkpoint inhibitors/agonists e.g. PD-1, PD-L1 inhibitors
- Antibody-dependent cellular cytotoxicity (ADCC) effects including evolution of cetuximab effect
- NK modulating agent testing
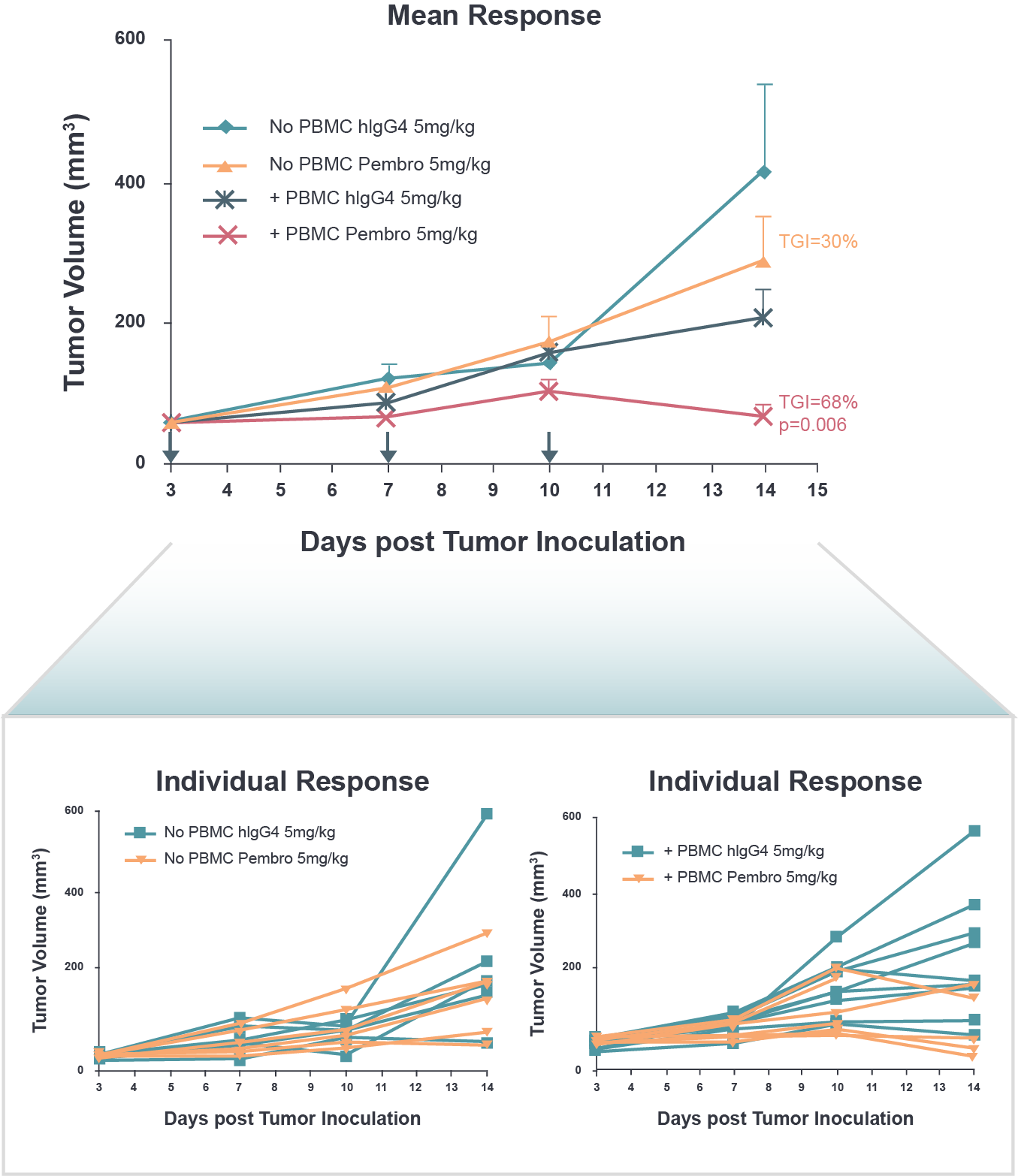
HCC827 NSCLC Model Tumor
Growth Inhibition Following Anti-PD-1 Treatment
